 Amongst these proteins are the χ subunit of the clamp loader 6– 11 and DnaG primase 12, 13. The tetrameric SSB protein not only protects the vulnerable single-stranded DNA and holds it in a proper conformation, but also engages in several interactions with proteins involved in DNA replication, repair and recombination via its highly conserved amphipathic C-terminus 5. At least two cores are required for the simultaneous replication of the two parental DNA strands 4. Depending on the number of τ proteins incorporated in the clamp loader complex, up to three cores can be contained in the pol III holoenzyme. The clamp loader not only loads the ring-shaped β 2 clamp onto the DNA, but also organises the whole replication fork, undergoing protein-protein interactions with the core, the helicase and SSB 1– 3. the β 2 sliding clamp that confers processivity to the core and 3. the αεθ core that comprises the 5′−3′ polymerase and the 3′−5′ proofreading activities, 2. Pol III is composed of three sub-complexes: 1. coli, the hexameric helicase (DnaB), primase (DnaG), single-stranded DNA-binding protein (SSB) and DNA polymerase III holoenzyme (pol III) as the replicative DNA polymerase are necessary for the progression of DNA synthesis. Several proteins and protein complexes have to cooperate at the bacterial replication fork in order to fulfil this demanding task. The exact replication of the genome is a prerequisite for the division of the bacterial cell. 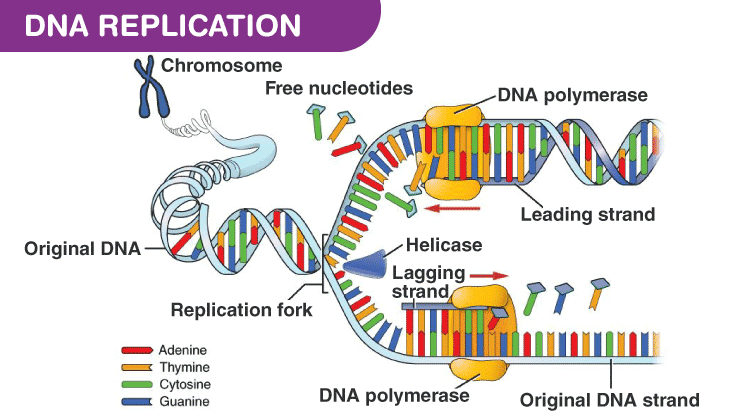 
The concurrent binding of primase and pol III is highly plausible, since even the SSB tetramer situated directly next to the 3′-terminus of the primer provides four C-termini for protein-protein interactions. About 10 nucleotides have to be added in order to displace one of the two primase molecules bound to SSB-saturated G4ori. Even pol III-mediated primer elongation by one or several DNA nucleotides does not result in the dissociation of DnaG. We found that the addition of pol III does not lead to a displacement of primase, but to the formation of higher complexes. Using a replication system containing the origin of replication of the single-stranded DNA phage G4 (G4ori) saturated with SSB, we tested whether DnaG and pol III can bind concurrently to the primed template. As both primase and the χ subunit of pol III interact with the highly conserved C-terminus of single-stranded DNA-binding protein (SSB), it had been proposed that the binding of both proteins to SSB is mutually exclusive. coli, a switch between DnaG primase and DNA polymerase III holoenzyme (pol III) activities has to occur every time when the synthesis of a new Okazaki fragment starts. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
